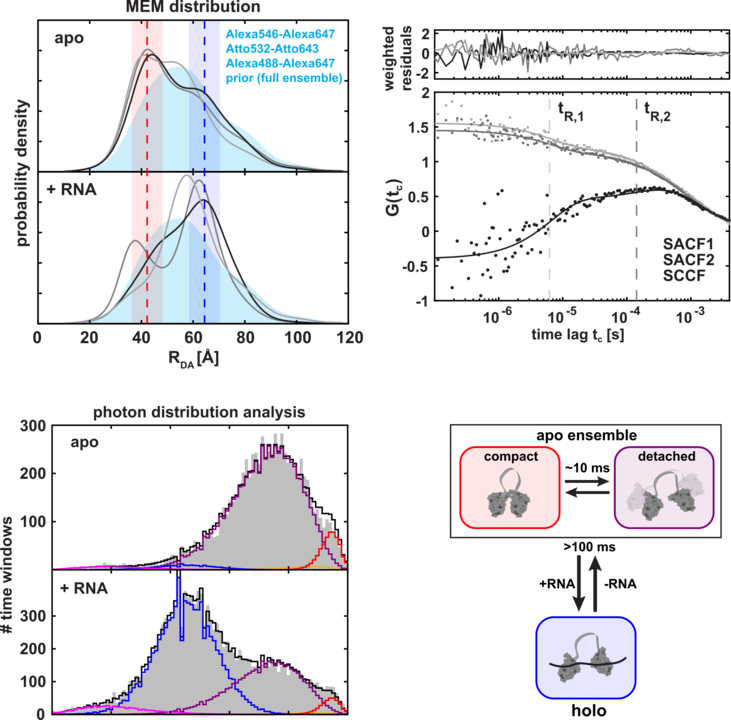
We are pleased to announce that our findings have now been officially published online, by Nature Methods! Alongside Prof. Don C. Lamb and Prof. Thorben Cordes from Ludwig Maximilian University of Munich, we have assessed the reliability and accuracy of single-molecule FRET studies in the international blind-study comprising 19 laboratories, using dynamic protein systems. Chosen benchmark systems were Maltose Binding Protein of ATP binding cassette, and a truncated version of splicing factor U2AF2, which vary in timescale as well the complexity of conformational dynamics. We have demonstrated the capacity of smFRET studies to reliably detect and quantify dynamics and associated structural changes, with the precision of ≤ 2Å and accuracy of ≤ 5Å. In the article, there is much beyond to read, with some of the highlights being practical discussions on setup designs, data quality and calibration uncertainty. We show that the error in the calibration, and in particular the error in γ parameter, is the leading factor that limits the precision of smFRET measurements. Thus, the precision is the highest for relative RET measurements (e.g. for distinct conditions (+/-) ligand). Furthermore, we addressed long-standing issues of detection and correction of dye sticking and we were able to determine the lower limit for the detection of small-scale distance fluctuations on the order of ≤ 5Å.
here you can find the article of the HHU: https://www.hhu.de/en/news-article/biomolecules-demonstrating-the-precision-of-optical-nanometer-measurements-for-size
Our Open Access article can be found under: https://www.nature.com/articles/s41592-023-01807-0
Agam, G., Gebhardt, C., Popara, M., Mächtel, R., Folz, J., ... , Barth, A., Seidel, C.A.M., Lamb, D.C. and Cordes, T., Reliability and accuracy of single-molecule FRET studies for characterization of structural dynamics and distances in proteins. Nat Methods (2023).